Fenbendazole and Cancer: What Current Case Reports and Research Really Suggest
Cancer affects countless individuals and families, often bringing anxiety, unanswered questions, and a strong desire to find anything that might help. When standard treatments seem insufficient or overwhelming, many people begin looking into unconventional options and newly discussed compounds. One name that has drawn growing interest is fenbendazole, a veterinary antiparasitic drug that has entered public conversation because of recent case reports and anecdotal patient stories.
So why is fenbendazole being discussed in cancer circles, and what does the evidence actually show? This article explores the science behind the interest, reviews the recent patient case series, and outlines the most important precautions for anyone exploring this subject. By the end, you’ll also find practical ways to discuss emerging research with your healthcare team.
What Is Fenbendazole?
Fenbendazole is part of the benzimidazole family of medications. It is commonly used in veterinary medicine to treat parasitic infections in animals such as dogs, horses, and farm livestock. In animal care, it is known for being relatively inexpensive, easy to obtain, and generally well tolerated.
Interest in fenbendazole has increased because some early laboratory research has suggested that it may do more than target parasites. Scientists have investigated whether it could also affect cancer cells under experimental conditions.
Researchers studying fenbendazole in preclinical settings, including cell-based and animal studies, have proposed several possible mechanisms:
- It may disrupt microtubules, which are essential for cell division.
- It may interfere with glucose metabolism, a process cancer cells often depend on heavily.
- It may trigger cellular stress responses that can damage or weaken tumor cells.
These findings help explain why repurposed drugs sometimes attract attention in oncology research. Still, laboratory results are only a starting point. What works in a dish or in animals does not automatically work safely or effectively in humans.
Why Fenbendazole Has Become a Topic in Cancer Discussions
The growing conversation around fenbendazole is tied to both science and personal stories. People facing advanced illness often investigate every possible option, especially when a treatment appears affordable and easy to access. Repurposed drugs are particularly appealing because they may already be familiar, widely available, and less expensive than newly developed therapies.
Fenbendazole fits that pattern. It is sold in veterinary formulations, and online communities have shared self-directed protocols based on personal experiences. Public interest has also been fueled by anecdotal accounts, including widely circulated stories such as Joe Tippens’ experience involving lung cancer and supplement use.
However, there is an important distinction to keep in mind: availability is not the same as proven clinical value. A product being easy to obtain does not mean it has been shown to be safe or effective for cancer treatment in humans.
Major cancer organizations, including the American Cancer Society, have emphasized that while similar antiparasitic compounds may show promise in early experiments, human evidence for fenbendazole remains limited.
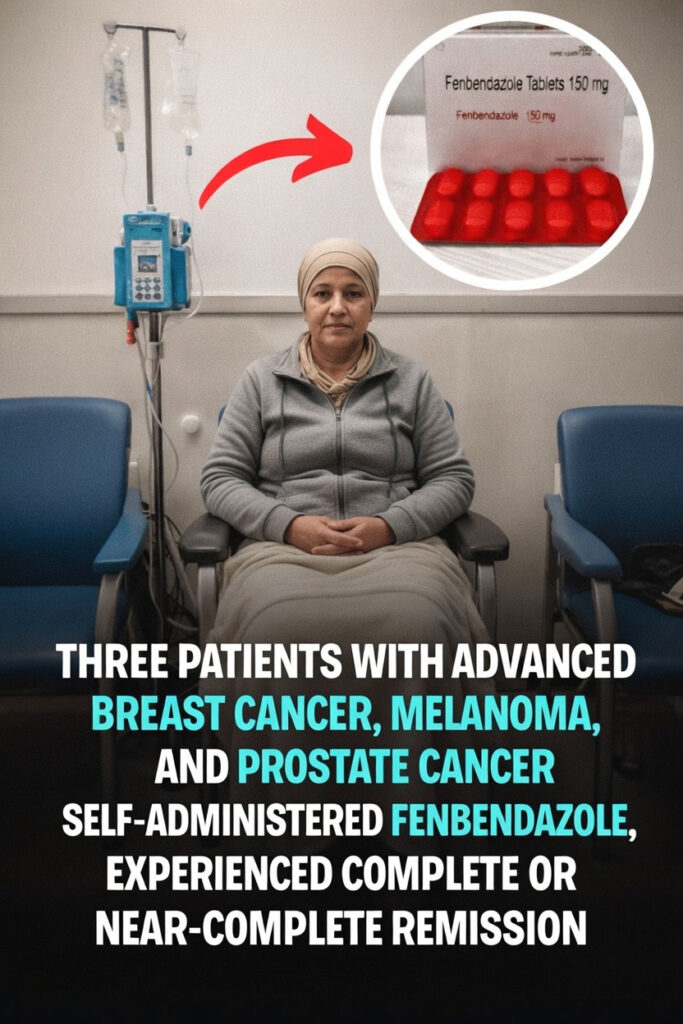
The 2025 Case Series: What the Three Reported Patients Experienced
A 2025 oncology case report described three people with advanced cancers who added fenbendazole to their routines after standard treatment experiences. The reported cases involved:
- Metastatic breast cancer
- Recurrent melanoma
- Advanced prostate cancer
According to the report, the patients described notable imaging outcomes:
- Two patients had scans showing a complete disappearance of detectable disease.
- One patient had an almost complete reduction in visible tumor burden.
- Follow-up periods ranged from roughly 11 months to nearly 3 years, with continued favorable imaging findings during that time.
- No major side effects were reported during the observation period.
These outcomes naturally sound encouraging. However, the authors themselves noted that these were case reports, not controlled clinical trials. That distinction matters a great deal.
Case reports can highlight unusual or promising outcomes, but they cannot prove cause and effect. There may be other explanations, including:
- Individual biological differences
- The impact of other supportive measures
- Variation in disease behavior
- Delayed effects from prior treatments
- Natural fluctuations in cancer progression
In the reported cases, fenbendazole was used along with other supportive strategies, while chemotherapy was not part of the described regimens. Because so many variables were involved, it is impossible to conclude from these cases alone that fenbendazole caused the reported responses.
What These Reports Mean — and What They Do Not Mean
The recent case series adds another layer to the discussion around drug repurposing in cancer care. It shows that some individual experiences have been striking enough to document in the medical literature. That alone makes the topic scientifically interesting.
Still, these reports do not establish fenbendazole as a proven cancer treatment.
To confirm whether a drug truly helps patients, researchers need:
- Large, well-designed clinical trials
- Clear dosing standards
- Safety monitoring
- Comparisons against standard care or placebo
- Long-term follow-up across diverse patient groups
Without those steps, strong conclusions cannot be made. Inspiring stories can generate valuable hypotheses, but they do not replace rigorous evidence.
Key Expert Concerns and Limitations
At this time, no large-scale clinical trials have confirmed a role for fenbendazole in routine human cancer treatment. That is one of the biggest reasons experts urge caution.
There are several additional concerns:
- Drug interactions: Fenbendazole could potentially interact with cancer therapies, prescription medications, or supplements.
- Product quality: Veterinary products are not intended or standardized for human cancer use.
- Unknown long-term risks: The effects of prolonged use in people are not well established.
- Delayed proven care: Self-treatment may lead some patients to postpone therapies with known benefits.
- Monitoring challenges: Without medical supervision, side effects or complications may go unnoticed.
Related drugs in the same benzimidazole class have received more attention in human medicine. For example, mebendazole, which is approved for treating parasitic infections in humans, has been studied more extensively in oncology settings. Even so, the results have been mixed, and it is still not considered standard cancer therapy.
Fenbendazole Compared With Similar Benzimidazole Compounds
To better understand the discussion, it helps to compare fenbendazole with related drugs often mentioned in repurposing research.
-
Fenbendazole
- Approved for veterinary use
- Limited human cancer data
- Recently highlighted in case reports and anecdotal discussions
-
Mebendazole
- Approved for human parasitic infections
- Studied more often in early oncology research
- Findings remain inconsistent and not yet practice-changing
-
Albendazole
- Belongs to the same drug class
- Occasionally examined in off-label contexts
- Not a standard treatment for cancer
Although these compounds may share possible laboratory mechanisms, they differ in human approval status, available research, and clinical relevance.

Practical Steps for Staying Informed and Protecting Your Health
If you are curious about fenbendazole or similar repurposed drugs, the best approach is one rooted in safety and reliable information. Here are several practical steps to consider:
1. Speak honestly with your doctor
Bring any articles, case reports, or stories you have found to your oncologist or healthcare provider. A good medical team can help you understand what is promising, what is uncertain, and what may be risky in your specific situation.
2. Build around evidence-based care
No matter what research topics interest you, your overall health still benefits from core supportive measures, such as:
- Good nutrition
- Appropriate physical activity
- Stress management
- Sleep support
- Following recommended treatment plans
These foundations are important during any cancer journey.
3. Use trustworthy sources
When researching emerging therapies, rely on credible information such as:
- Peer-reviewed medical journals
- Major cancer organizations
- University medical centers
- Clinical trial databases
Be cautious with social media claims or forum-based protocols that lack medical oversight.
4. Ask about clinical trials
If drug repurposing interests you, ask whether there are any clinical trials studying repurposed agents, novel combinations, or related compounds. Trial participation may offer access to structured monitoring while also helping advance scientific understanding.
5. Track symptoms and treatment responses
Keep a written record of:
- Symptoms
- Energy levels
- Side effects
- Scan results
- Medication or supplement use
Accurate tracking can help your healthcare team make safer, better-informed decisions.
Balancing Hope With Scientific Caution
Cancer research continues to evolve, and stories of unexpected outcomes remind us that medicine still has much to discover. The recent fenbendazole case series contributes to an ongoing conversation about whether older, non-cancer drugs might someday play a role in oncology.
That said, curiosity should always be paired with caution. At present, fenbendazole remains an investigational topic in this context, not a validated cancer treatment. Real progress requires carefully controlled human studies that can determine whether a drug is both safe and beneficial for larger groups of patients.
If this subject interests you, let it be a reason to ask thoughtful questions and have open conversations with qualified healthcare professionals. They are best equipped to help you weigh emerging research against your individual diagnosis, treatment history, and health needs.
FAQ
What is fenbendazole primarily used for?
Fenbendazole is an antiparasitic medication mainly used in animals to treat worms and other parasitic infections. It is not approved for human medical use in cancer treatment.
Are there human studies on fenbendazole for cancer?
Most of the current evidence comes from preclinical research such as laboratory and animal studies, along with a small number of case reports. There are no large randomized clinical trials proving its safety or effectiveness for cancer in humans.
Should I talk to my oncologist about alternative or emerging options?
Yes. You should always discuss any alternative, off-label, or experimental approach with your oncologist. Open communication helps ensure safer care and allows your doctor to give advice tailored to your condition.