A New Direction in Parkinson’s Care: Understanding Tavapadon
Living with Parkinson’s disease can turn everyday activities into major challenges. Stiffness, tremors, and slowed movement often interfere with independence and reduce quality of life for millions of people around the world. Still, there is reason for optimism. Scientists are developing new ways to improve dopamine signaling in the brain, and one emerging therapy now under FDA review is tavapadon.
This article explains what makes tavapadon unique, how it could fit into Parkinson’s treatment strategies, and what practical conversations you can have with your healthcare team.
Why Current Parkinson’s Treatments Can Be Difficult
Parkinson’s disease occurs when the brain gradually loses cells that produce dopamine, a chemical essential for smooth and coordinated movement. For decades, levodopa has been one of the most important treatments because it helps restore dopamine levels. Many patients also take dopamine agonists, which imitate dopamine by attaching to dopamine receptors.
Even though these treatments can be effective, they also have limitations.
- Levodopa often needs to be taken several times a day.
- Over time, it may lead to motor fluctuations, meaning symptom control can vary throughout the day.
- Patients may experience periods of better movement, known as “on” time, mixed with “off” periods or involuntary movements called dyskinesia.
- Traditional dopamine agonists, which mainly affect D2 and D3 receptors, can sometimes cause side effects such as:
- excessive daytime sleepiness
- impulse control problems
- swelling in the legs
This raises an important question: could a treatment target movement-related dopamine pathways more precisely while possibly avoiding some of these unwanted effects?
How Tavapadon Works
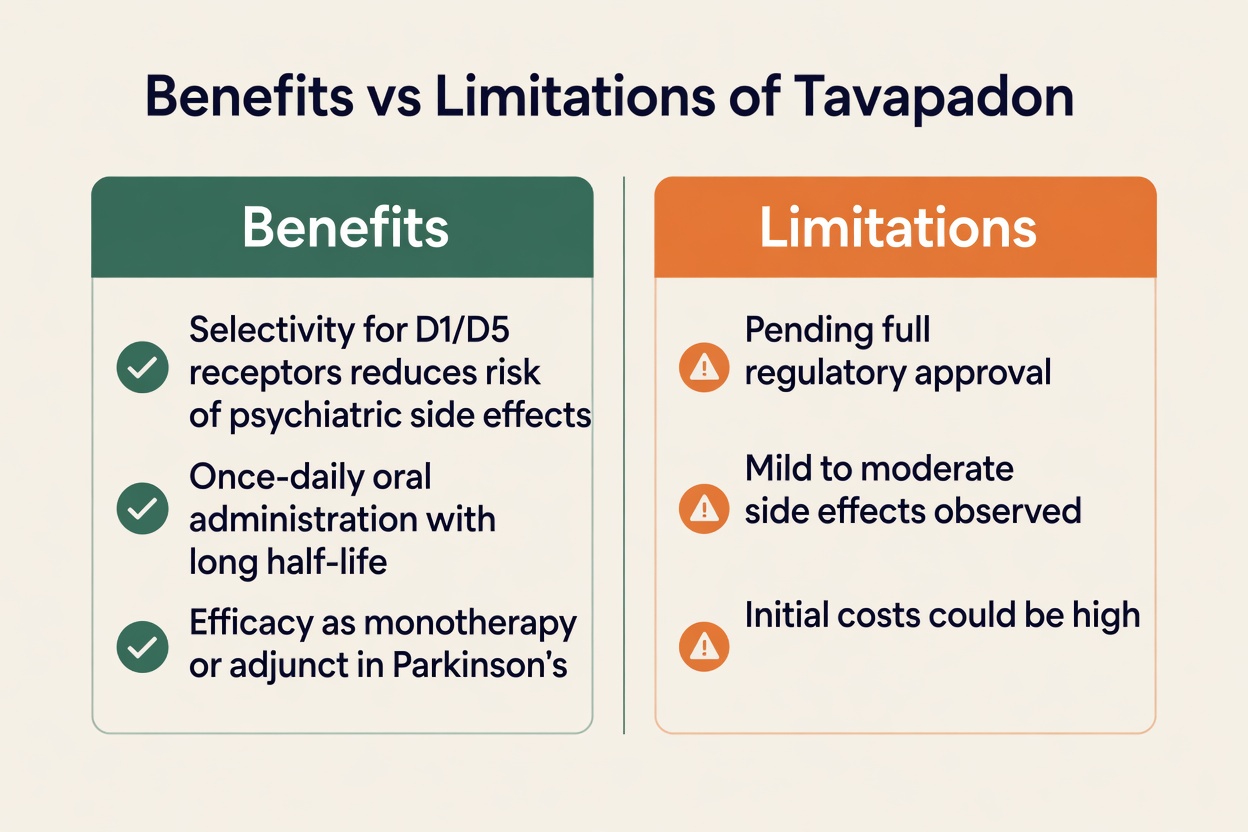
Tavapadon is an investigational once-daily oral medication designed as a selective partial agonist of the D1 and D5 dopamine receptors. That sets it apart from many currently used dopamine agonists, which mostly act on D2 and D3 receptors.
The D1/D5 receptor family is closely tied to the brain’s direct pathway, a system involved in starting and smoothing movement. By focusing on this pathway, tavapadon is intended to support motor function in a more targeted way.
A helpful way to think about it is this: the brain uses several dopamine “switches.” Older dopamine agonists tend to activate multiple switches at once, which may help symptoms but can also overstimulate certain areas. Tavapadon is designed to work more selectively and more gently because it is a partial agonist, meaning it provides a controlled signal rather than maximum stimulation.
What the TEMPO Studies Found
Tavapadon has been evaluated in the TEMPO clinical trial program, including TEMPO-1, TEMPO-2, and TEMPO-3. These studies examined the drug in both early-stage Parkinson’s disease and in people with more advanced disease already using levodopa.
The results were encouraging:
- In some participants, tavapadon improved motor function scores.
- When used together with levodopa, it increased daily “on” time.
- Some people gained about one additional hour of good symptom control per day.
- Many reported side effects were described as mild to moderate.
Another notable point is that trial data suggested lower rates of some side effects commonly linked to D2/D3-targeting drugs, such as:
- impulse control disorders
- excessive sleepiness
That said, responses can differ from person to person, and broader real-world experience will be needed to fully understand its long-term effect profile.
Key Clinical Trial Highlights
Across the TEMPO program, researchers reported several consistent findings.
Early Parkinson’s Disease
In TEMPO-1 and TEMPO-2, tavapadon was tested as a standalone treatment in people with early Parkinson’s disease. Compared with placebo, it produced meaningful improvements in:
- movement-related symptoms
- daily functioning
- standardized Parkinson’s rating scale scores
Parkinson’s With Motor Fluctuations
In TEMPO-3, researchers looked at people already taking levodopa who were experiencing motor fluctuations. Adding tavapadon led to:
- more “on” time without troublesome dyskinesia
- less “off” time
Longer-Term Follow-Up
Follow-up data over additional months suggested that many participants maintained benefit over time, and the overall safety profile was considered generally favorable by investigators.
Reported Side Effects
The most commonly reported side effects in studies included:
- nausea
- headache
- dizziness
Most of these were mild to moderate. Serious adverse events occurred at rates that were similar to, or in some cases slightly higher than, placebo in certain trials. Importantly, hallucinations and impulse control problems appeared comparable to placebo in the available data, which fits with tavapadon’s more selective receptor targeting.
Comparing Tavapadon With Other Parkinson’s Medications
Here is a simple educational comparison:
-
Traditional dopamine agonists
- Mostly target D2/D3 receptors
- May improve motor symptoms
- Can sometimes be linked to sleepiness, compulsive behaviors, or leg swelling
-
Tavapadon
- Selective D1/D5 partial agonist
- Designed to support movement through the brain’s direct pathway
- Taken once daily
- May have fewer D2/D3-related side effects based on trial findings
-
Levodopa
- Remains highly effective for many patients
- Often requires multiple doses per day
- Can contribute to motor fluctuations over time
This comparison is for general education only. The right treatment plan depends on your symptoms, stage of disease, other health conditions, and your doctor’s guidance.
Who May Want to Ask About Tavapadon?
Tavapadon has been studied in adults with:
- early Parkinson’s disease as an initial treatment option
- more advanced Parkinson’s disease as an add-on to levodopa
Because it is designed for once-daily dosing, it may be especially interesting to people who want a simpler medication routine or who struggle with symptom fluctuations during the day.
However, it is important to remember that tavapadon is not yet FDA-approved based on the latest available information. AbbVie submitted a New Drug Application in 2025, supported by the TEMPO trial data. FDA review can take several months, so availability will depend on the final regulatory decision.
Practical Steps You Can Take Right Now
While waiting for new Parkinson’s treatments to become available, there are several useful actions you can start today.
1. Keep a Daily Symptom Log
Use a notebook or mobile app to record:
- medication times
- “on” and “off” periods
- tremor, stiffness, or slowness
- how meals or exercise affect symptoms
Bringing this information to appointments can help your neurologist make better treatment decisions.
2. Review Your Medication Schedule
Ask your healthcare provider whether changing the timing or combination of your medications could reduce fluctuations and improve daily function.
3. Stay Physically Active
Gentle movement can support mobility and confidence. Consider:
- short daily walks
- stretching
- balance exercises
- physical therapy routines
Even 10 to 15 minutes a day can make a meaningful difference.
4. Follow Trusted Parkinson’s Resources
Reliable organizations can help you stay informed about clinical updates and emerging treatments, including:
- The Michael J. Fox Foundation
- American Parkinson Disease Association
5. Prepare Questions for Your Next Appointment
Useful questions may include:
- “Could newer therapies such as selective dopamine agonists be relevant for me?”
- “How can I better manage my current ‘off’ periods?”
- “Are there lifestyle changes that could improve my symptom control?”
These steps may seem small, but they can help you feel more informed and more involved in your care.
What Tavapadon May Mean for the Future
The development of tavapadon reflects a larger shift in Parkinson’s treatment: moving toward more precise dopamine modulation. Instead of broadly stimulating several receptor pathways, researchers are studying whether targeting D1/D5 receptors can provide strong motor support with practical once-daily use.
As more data become available, including findings from ongoing research such as TEMPO-4, experts should gain a clearer understanding of where tavapadon may fit in future Parkinson’s care.
For many people living with Parkinson’s disease, this type of targeted strategy offers real hope for better daily symptom control without adding more complexity to treatment.
Frequently Asked Questions
How is tavapadon different from other Parkinson’s drugs?
Tavapadon works as a selective partial agonist at D1 and D5 dopamine receptors. Many currently used dopamine agonists mainly target D2 and D3 receptors. Researchers are studying whether this difference could improve movement symptoms while influencing side effects in a different way.
Is tavapadon available now?
No. Tavapadon is still considered investigational. The FDA is reviewing the New Drug Application submitted in 2025, and official decisions about approval and availability have not yet been finalized.
What should I talk about with my doctor regarding new Parkinson’s therapies?
Discuss:
- your current symptom patterns
- any side effects from present medications
- whether once-daily treatment options might suit your routine
- your experience with “on” and “off” time
Your healthcare team can explain how new and emerging therapies may or may not fit into your treatment plan.